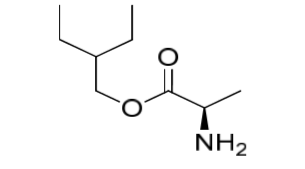
Remdesivir Impurity 9
| Product Name | Remdesivir Impurity 9 |
|---|---|
| Alternate Names | Remdesivir Impurities, Impurities of Remdesivir |
| CAT No. | CS-O-35700 |
| CAS No. | 1689993-65-4 |
| Category | Impurities |
| Stock | IN-Stock |
| Mol. Wt. | 173.26 g/mol |
| Mol. For. | C9H19NO2 |
| Hazardous | This is not a Hazardous Compound |
| COA | View Sample COA |
| MSDS | View Sample MSDS |
| Parent API | Remdesivir |
| Smileys | O=C1C(N2CCOCC2)=CC=CN1C3=CC=C([N+]([O-])=O)C=C3 |
| Controlled | No |
| Shipping | Free for purchase above 1000$ |
| Delivery | In-Stock, products will be dispatched within 24 hours via FedEx for USA, Europe, and other countries. |
| Return | Returns/replacement accepted if you are not satisfied with the quality of the product, (please send us an email with the reason/issues which are facing, within 15 days, after receipt of the product). |
| Ordering | Place your order online or by email sales@clearsynth.com |
Remdesivir Impurity 9 is a chemical compound that is commonly used as an impurity reference standard in laboratories. It is a white to off-white powder that is soluble in organic solvents such as methanol, ethanol, and chloroform. The chemical formula for Remdesivir Impurity 9 is C22H25N5O5 and its molecular weight is 451.46.
Remdesivir Impurity 9 is often used in the pharmaceutical industry as a reference standard to identify and quantify impurities in Remdesivir drug substances and finished dosage forms. It is also used in the development and validation of analytical methods for determining the purity and quality of Remdesivir.
The purity of Remdesivir Impurity 9 is typically measured using high-performance liquid chromatography (HPLC) or gas chromatography (GC). These analytical techniques separate the individual components of a mixture based on their chemical properties and provide a quantitative measure of the purity of the sample.
In conclusion, Remdesivir Impurity 9 is an important reference standard in the pharmaceutical industry, used to ensure the purity and quality of Remdesivir drug substances and finished dosage forms. Its chemical properties and analytical methods make it a valuable tool for drug development and quality control.
Get an Instant Quote
Related Compounds
Remdesivir Desphenyl Acid Impurity | NSJ-Remdesivir-RS012 | Remdesivir Impurity 14 | Remdesivir Impurity 11 | L-Alanine, N-(chlorophenoxyphosphinyl)-, 2-ethylbutyl ester (IMP 7) | Remdesivir Phosphate Impurity | Remdesivir Impurity 16 | NSJ-Remdesivir-RS004 | GS-773095 | Remdesivir Impurity 3 | Remdesivir Impurity 16 | Remdesivir Impurity 13 | Remdesivir Impurity 15 | Remdesivir Desphenyl Impurity | Remdesivir Impurity 10 | L-Alanine, N-[(R)-(4-nitrophenoxy)phenoxyphosphinyl]-, 2-ethylbutyl ester (IMP 5) | Remdesivir Impurity 17 | D-Alanine, N-[(S)-(4-nitrophenoxy)phenoxyphosphinyl]-, 2-ethylbutyl ester (IMP 6) | Remdesivir R-P-D Alanine Isomer | Remdesivir Bis Phosphoryl impurity | Remdesivir Impurity 2 | Remdesivir impurity-18 Triethylamine salt | L-Alanine, N-[(S)-(4-nitrophenoxy)phenoxyphosphinyl]-, 2-ethylbutyl ester (IMP 4) | Remdesivir Impurity 18 | Remdesivir Impurity C | Remdesivir Nucleoside Monophosphate Ammonium salt | NSJ-Remdesivir-RS005 | Remdesivir Impurity 8 | Remdesivir N-oxide | Remdesivir R-P isomer and S-isomer at CN | Remdesivir Impurity 1 | Remdesivir Impurity 41 | Remdesivir Impurity 43 | NSJ-Remdesivir-RS009 | Remdesivir Impurity 12 |