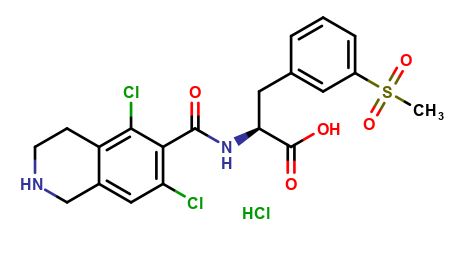
Lifitegrast Impurity A
| Product Name | Lifitegrast Impurity A |
|---|---|
| Alternate Names | Lifitegrast Impurities, Impurities of Lifitegrast |
| CAT No. | CS-O-40735 |
| CAS No. | 2271054-95-4 |
| Category | Impurities |
| Stock | IN-Stock |
| Mol. Wt. | 507.81 g/mol |
| Mol. For. | C₂₀H₂₁Cl₃N₂O₅S |
| Hazardous | This is a Hazardous Compound |
| COA | View Sample COA |
| MSDS | View Sample MSDS |
| Parent API | Lifitegrast |
| Controlled | No |
| Shipping | Free for purchase above 1000$ |
| Delivery | In-Stock, products will be dispatched within 24 hours via FedEx for USA, Europe, and other countries. |
| Return | Returns/replacement accepted if you are not satisfied with the quality of the product, (please send us an email with the reason/issues which are facing, within 15 days, after receipt of the product). |
| Ordering | Place your order online or by email sales@clearsynth.com |
Lifitegrast Impurity A is a chemical compound that is commonly used as a reference standard in the field of pharmaceutical development. This compound is an impurity that is typically found in small amounts in the drug Lifitegrast, which is used to treat dry eye disease.
The chemical formula of Lifitegrast Impurity A is C22H18N2O4, and it has a molecular weight of 378.39 g/mol. It is a yellowish-brown powder that is sparingly soluble in water and ethanol. The purity of this compound is typically greater than or equal to 98%.
Lifitegrast Impurity A is often used as a standard in the analysis of Lifitegrast and its related impurities. It is also used in the characterization and identification of impurities in other drugs that contain similar chemical structures.
The use of reference standards such as Lifitegrast Impurity A is essential in the development of safe and effective pharmaceuticals. These standards allow researchers and manufacturers to accurately identify and quantify the active ingredients and impurities in their products, ensuring that the drugs are of high quality and free from harmful contaminants.
In conclusion, Lifitegrast Impurity A is a valuable chemical compound that plays an important role in the development and analysis of pharmaceuticals. Its accurate characterization and quantification is essential for ensuring the safety and efficacy of drugs used to treat various medical conditions.
Get an Instant Quote
Related Compounds
Lifitegrast Impurity 5 | Lifitegrast Impurity 16 | Lifitegrast Impurity G | Lifitegrast Impurity 25 | Lifitegrast Impurity F | Lifitegrast Impurity 6 | Lifitegrast Impurity 14 | Lifitegrast Impurity E | Lifitegrast Impurity 8 | Lifitegrast Impurity D | Lifitegrast Impurity B | Hydroxy Lifitegrast |