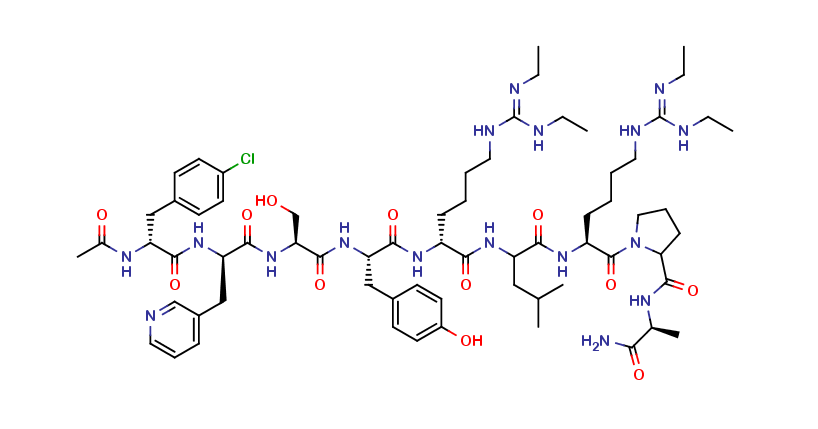
Ganirelix Impurity A
| Product Name | Ganirelix Impurity A |
|---|---|
| Alternate Names | Ganirelix Impurities, Impurities of Ganirelix |
| CAT No. | CS-P-01379 |
| CAS No. | Not Available |
| Category | Impurities |
| Stock | IN-Stock |
| Mol. Wt. | 1373.08 g/mol |
| Mol. For. | C₆₇H₁₀₂ClN₁₇O₁₂ |
| Hazardous | This is a Hazardous Compound |
| COA | View Sample COA |
| MSDS | View Sample MSDS |
| Parent API | Ganirelix |
| Purity | 95% |
| Therapeutic | Synthetic Peptides |
| Controlled | No |
| Shipping | Free for purchase above 1000$ |
| Delivery | In-Stock, products will be dispatched within 24 hours via FedEx for USA, Europe, and other countries. |
| Return | Returns/replacement accepted if you are not satisfied with the quality of the product, (please send us an email with the reason/issues which are facing, within 15 days, after receipt of the product). |
| Ordering | Place your order online or by email sales@clearsynth.com |
Ganirelix Impurity A is a chemical compound that is commonly used in the pharmaceutical industry as a reference standard for the development and testing of drugs. This impurity is a byproduct of the synthesis of ganirelix, which is a medication used in the treatment of infertility. Ganirelix Impurity A is a synthetic peptide that is structurally similar to ganirelix, but differs in its chemical composition.
The chemical information of Ganirelix Impurity A includes its molecular formula, molecular weight, and purity. Its molecular formula is C63H88N14O14S, and its molecular weight is 1294.62 g/mol. The purity of Ganirelix Impurity A is typically measured by high-performance liquid chromatography (HPLC), which is a common analytical technique used in the pharmaceutical industry.
In terms of usage, Ganirelix Impurity A is primarily used as a reference standard for the quality control of ganirelix. It is also used in the development and testing of other drugs that are structurally similar to ganirelix. Additionally, Ganirelix Impurity A can be used as a research tool in the study of peptide chemistry and biochemistry.
Overall, Ganirelix Impurity A plays an important role in the pharmaceutical industry as a reference standard, research tool, and quality control agent for the development and testing of drugs.
Get an Instant Quote
Related Compounds
Ganirelix Impurity E | Ganirelix Impurity C | Ganirelix Acetate Impurity B | Ganirelix Impurity F | Ganirelix Impurity B | Ganirelix Impurity D (Mono-Acetyl- Ganilelix) |