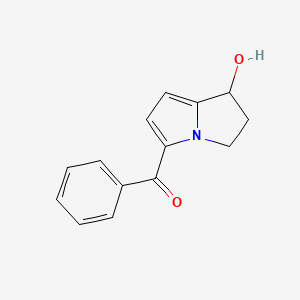
Ketorolac EP Impurity A
| Product Name | Ketorolac EP Impurity A |
|---|---|
| Alternate Names | Ketorolac Impurities, Impurities of Ketorolac |
| CAT No. | CS-T-15819 |
| CAS No. | 154476-25-2 |
| Category | Impurities |
| Stock | IN-Stock |
| Mol. Wt. | 227.26 g/mol |
| Mol. For. | C₁₄H₁₃NO₂ |
| Hazardous | This is a Hazardous Compound |
| COA | View Sample COA |
| MSDS | View Sample MSDS |
| Parent API | Ketorolac |
| Purity | 95% |
| Therapeutic | Anti-Migraines |
| Smileys | C1CN2C(=CC=C2C(=O)C3=CC=CC=C3)C1O |
| Controlled | No |
| Shipping | Free for purchase above 1000$ |
| Delivery | In-Stock, products will be dispatched within 24 hours via FedEx for USA, Europe, and other countries. |
| Return | Returns/replacement accepted if you are not satisfied with the quality of the product, (please send us an email with the reason/issues which are facing, within 15 days, after receipt of the product). |
| Ordering | Place your order online or by email sales@clearsynth.com |
Ketorolac EP Impurity A is a chemical byproduct that is commonly found in the manufacturing process of ketorolac, a nonsteroidal anti-inflammatory drug (NSAID) used to treat pain and inflammation. This impurity is a known metabolite of ketorolac, which means that it is produced when the drug is broken down in the body.
Although Ketorolac EP Impurity A is not intended for human consumption, it is important to understand its chemical properties and potential effects. The impurity has a molecular weight of 255.21 g/mol and a chemical formula of C14H13NO3. It is a white to off-white crystalline powder that is soluble in organic solvents such as methanol and ethyl acetate.
In terms of usage, Ketorolac EP Impurity A is primarily used by pharmaceutical companies as a reference standard for quality control purposes. By testing for the presence and amount of this impurity in their ketorolac products, manufacturers can ensure that their drugs are safe and effective for human use.
It is worth noting that Ketorolac EP Impurity A has been linked to potential health risks in some studies. For example, a 2017 study published in the Journal of Pharmaceutical and Biomedical Analysis found that the impurity may be genotoxic, meaning that it has the potential to damage DNA and increase the risk of cancer. However, further research is needed to fully understand the implications of this finding.
Get an Instant Quote
Related Compounds
Ketorolac 1-Keto Analog | Bromo ketorolac | Ketorolac EP Impurity B | Ketorolac EP Impurity H | Ketorolac EP Impurity J | Ketorolac EP Impurity D | Ketorolac EP Impurity C | Ketorolac EP Impurity F | Ketorolac tromethamine | Ketorolac EP Impurity I | N-Nitroso Ketorolac 2-Benzoylpyrrole Impurity | N-Nitroso Ketorolac Triethylester Intermediate | Ketorolac EP Impurity E | N-(1,3-dihydroxy-2-(hydroxymethyl)propan-2-yl)-N-hydroxynitrous amide | N-(1,3-dihydroxy-2-(hydroxymethyl)propan-2-yl)-N-methylnitrous amide | Ketorolac EP Impurity G |