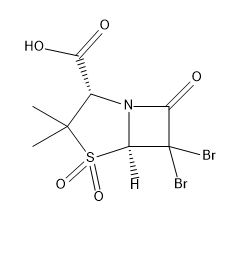
Sulbactam Related Compound E
| Product Name | Sulbactam Related Compound E |
|---|---|
| Alternate Names | Sulbactam Impurities, Impurities of Sulbactam |
| CAT No. | CS-T-17293 |
| CAS No. | 76646-91-8 |
| Category | Impurities |
| Stock | IN-Stock |
| Mol. Wt. | 391.03 g/mol |
| Mol. For. | C₈H₉Br₂NO₅S |
| Hazardous | This is a Hazardous Compound |
| COA | View Sample COA |
| MSDS | View Sample MSDS |
| Parent API | Sulbactam |
| Smileys | CC1(C(N2C(S1(=O)=O)C(C2=O)(Br)Br)C(=O)O)C |
| Controlled | No |
| Shipping | Free for purchase above 1000$ |
| Delivery | In-Stock, products will be dispatched within 24 hours via FedEx for USA, Europe, and other countries. |
| Return | Returns/replacement accepted if you are not satisfied with the quality of the product, (please send us an email with the reason/issues which are facing, within 15 days, after receipt of the product). |
| Ordering | Place your order online or by email sales@clearsynth.com |
Sulbactam Related Compound E is a chemical compound that is commonly used in the pharmaceutical industry as a reference standard for testing and quality control purposes. It is a derivative of Sulbactam, which is a beta-lactamase inhibitor used in combination with various antibiotics to treat bacterial infections.
Sulbactam Related Compound E is a white to off-white crystalline substance that has a molecular weight of 267.3 g/mol. It is soluble in water, methanol, and dimethyl sulfoxide (DMSO), and has a melting point of 206-208°C. The chemical structure of Sulbactam Related Compound E is similar to Sulbactam, with the addition of an ethyl group at the C-2 position of the thiazolidine ring.
In the pharmaceutical industry, Sulbactam Related Compound E is primarily used as a reference standard for the analysis of Sulbactam in drug formulations. It is also used in the development and validation of analytical methods for the determination of Sulbactam in various biological samples, including plasma and urine.
Due to its important role in the pharmaceutical industry, Sulbactam Related Compound E is subject to rigorous quality control measures to ensure its purity and identity. It is typically analyzed using high-performance liquid chromatography (HPLC), which allows for the accurate quantification of the compound in drug formulations and biological samples.
Get an Instant Quote
Related Compounds
Sulbactam EP Impurity C | Sulbactam Sodium EP Impurity C | Sulbactam Sodium EP Impurity G | Sulbactam Sodium EP Impurity D | Sulbactam EP Impurity D | Sulbactam Sodium EP Impurity A | Sulbactam Acid | Sulbactam Sodium EP Impurity F |