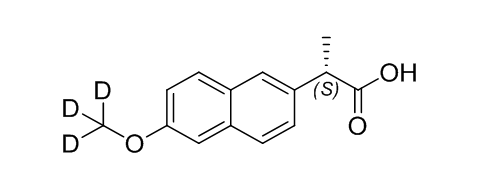
(S)-Naproxen D3
| Product Name | (S)-Naproxen D3 |
|---|---|
| Alternate Names | Naproxen Stable Isotopes, Stable Isotopes of Naproxen |
| CAT No. | CS-T-59127 |
| CAS No. | 1094102-82-5 |
| Category | Stable Isotopes |
| Stock | IN-Stock |
| Mol. Wt. | 233.28 g/mol |
| Mol. For. | C₁₄H₁₁D₃O₃ |
| Hazardous | This is not a Hazardous Compound |
| COA | View Sample COA |
| MSDS | View Sample MSDS |
| Parent API | Naproxen |
| Smileys | C[C@@H](C1=CC(C=CC(OC([2H])([2H])[2H])=C2)=C2C=C1)C(O)=O |
| Controlled | No |
| Shipping | Free for purchase above 1000$ |
| Delivery | In-Stock, products will be dispatched within 24 hours via FedEx for USA, Europe, and other countries. |
| Return | Returns/replacement accepted if you are not satisfied with the quality of the product, (please send us an email with the reason/issues which are facing, within 15 days, after receipt of the product). |
| Ordering | Place your order online or by email sales@clearsynth.com |
(S)-Naproxen D3 is a deuterated form of the nonsteroidal anti-inflammatory drug (NSAID) naproxen. It is commonly used for the treatment of pain, inflammation and fever. The deuterated form is used in scientific research and clinical trials as a tracer molecule for pharmacokinetic studies.
(S)-Naproxen D3 works by inhibiting the production of prostaglandins, which are responsible for causing pain, inflammation, and fever. It achieves this by inhibiting the activity of cyclooxygenase (COX) enzymes, which are involved in the production of prostaglandins. The deuterated form of naproxen has a slightly different chemical structure compared to the non-deuterated form, which makes it useful for pharmacokinetic studies.
The molecular formula of (S)-Naproxen D3 is C14H10D3NO3 and its molecular weight is 253.29 g/mol. It is a white or off-white crystalline powder that is slightly soluble in water. The compound has a melting point of 148-152 °C and has a purity of at least 98%.
Overall, (S)-Naproxen D3 is a useful tool for studying the pharmacokinetics of naproxen and can help researchers better understand how the drug is metabolized and eliminated from the body. Its chemical structure and properties make it an important tool for drug development and research.
Get an Instant Quote
Related Compounds
Rac Naproxen D3 Ethyl ester | Naproxen 13CD3 | rac O-Desmethyl Naproxen D3 | rac Naproxen-d3 Methyl Ester | Naproxen-1,3,4,5,7,8-d6 | rac-5-Bromo Naproxen-13CD3 | Rac-Naproxen D3 (Methoxy-D3) |