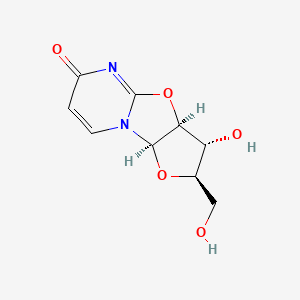
2,2-O-Cyclouridine
| Product Name | 2,2-O-Cyclouridine |
|---|---|
| Alternate Names | Uridine Impurities, Impurities of Uridine |
| CAT No. | CS-DA-00552 |
| CAS No. | 3736-77-4 |
| Category | Impurities |
| Stock | IN-Stock |
| Mol. Wt. | 226.19 g/mol |
| Mol. For. | C₉H₁₀N₂O₅ |
| Hazardous | This is a Hazardous Compound |
| COA | View Sample COA |
| MSDS | View Sample MSDS |
| Parent API | Uridine |
| Canonical Smiles | C1=CN2C3C(C(C(O3)CO)O)OC2=NC1=O |
| InchIKey | UUGITDASWNOAGG-CCXZUQQUSA-N |
| Inchl | InChI=1S/C9H10N2O5/c12-3-4-6(14)7-8(15-4)11-2-1-5(13)10-9(11)16-7/h1-2,4,6-8,12,14H,3H2/t4-,6-,7+,8-/m1/s1 |
| IUPAC | (2R,4R,5R,6S)-5-hydroxy-4-(hydroxymethyl)-3,7-dioxa-1,9-diazatricyclo[6.4.0.02,6]dodeca-8,11-dien-10-one |
| Controlled | No |
| Shipping | Free for purchase above 1000$ |
| Delivery | In-Stock, products will be dispatched within 24 hours via FedEx for USA, Europe, and other countries. |
| Return | Returns/replacement accepted if you are not satisfied with the quality of the product, (please send us an email with the reason/issues which are facing, within 15 days, after receipt of the product). |
| Ordering | Place your order online or by email sales@clearsynth.com |
2,2-O-Cyclouridine is a modified nucleoside that has gained interest in recent years due to its potential use in the development of nucleic acid-based therapeutics. This molecule consists of a uridine base that has been cyclized at the 2' and 2'' positions through the formation of an ether linkage, resulting in a unique cyclic structure.
One of the key applications of 2,2-O-Cyclouridine is in the development of antisense oligonucleotides (ASOs). ASOs are short, synthetic nucleic acid sequences that can bind to specific target RNA molecules and modulate their activity. By incorporating 2,2-O-Cyclouridine into ASOs, researchers have demonstrated improved binding affinity, nuclease resistance, and cellular uptake compared to traditional oligonucleotides.
In addition to its use in ASOs, 2,2-O-Cyclouridine has also been investigated for its potential as a prodrug. Prodrugs are inactive compounds that can be converted into active drugs within the body, usually through enzymatic or chemical reactions. In the case of 2,2-O-Cyclouridine, it can be converted into a potent antiviral agent known as 2'-C-methylcytidine. This prodrug approach has shown promise in the treatment of viral infections such as hepatitis C and respiratory syncytial virus.
From a chemical standpoint, 2,2-O-Cyclouridine is a stable molecule that is resistant to hydrolysis and enzymatic degradation. Its unique cyclic structure also confers increased rigidity and conformational stability compared to linear nucleosides. These properties make it an attractive building block for the development of nucleic acid-based drugs and therapeutics.
Get an Instant Quote
Related Compounds
Uridine Diphosphate-GlcNaz | ent-Idoxuridine | Uridine-5-diphosphocholine sodium salt | Uridine 5'-Monophosphate Disodium Salt | S4-(2-Cyanoethyl)-4-thio-2’-deoxyuridine | Doxifluridine Related compound A | Uridine 5'-Diphosphate Sodium Salt | Uridine-5'-triphosphate trisodium salt | Uridine Diphosphate Choline Ammonium Salt | Trifluridine Impurity 1 | Uridine 5-Tetraphosphate | Uridine Related Compound 1 | Uridine 3'-Monophosphate Disodium Salt | Fialuridine 5'-Monophosphate |