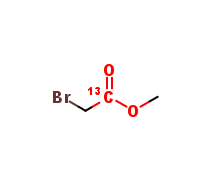
Acetic-1-13C-2-bromoacid-methylester
| Product Name | Acetic-1-13C-2-bromoacid-methylester |
|---|---|
| Alternate Names | Bromoacid Stable Isotopes, Stable Isotopes of Bromoacid |
| CAT No. | CS-BX-00830 |
| CAS No. | 62081-18-9 |
| Category | Stable Isotopes |
| Stock | Enquire |
| Mol. Wt. | 153.97 g/mol |
| Mol. For. | 13CC2H5BrO2 |
| Hazardous | This is not a Hazardous Compound |
| COA | View Sample COA |
| MSDS | View Sample MSDS |
| Parent API | Bromoacid |
| Controlled | No |
| Shipping | Free for purchase above 1000$ |
| Delivery | In-Stock, products will be dispatched within 24 hours via FedEx for USA, Europe, and other countries. |
| Return | Returns/replacement accepted if you are not satisfied with the quality of the product, (please send us an email with the reason/issues which are facing, within 15 days, after receipt of the product). |
| Ordering | Place your order online or by email sales@clearsynth.com |
Acetic-1-13C-2-bromoacid-methylester is a chemical compound that is widely used in organic chemical synthesis as a building block for the preparation of more complex chemical compounds. The compound is a derivative of acetic acid, which is a colorless liquid that has a pungent smell and is widely used in the food industry as a preservative and flavoring agent.
Acetic-1-13C-2-bromoacid-methylester contains a 13C isotope, which is a stable isotope of carbon that is commonly used in nuclear magnetic resonance (NMR) spectroscopy for structural analysis of organic molecules. The compound is also a methyl ester, which makes it a good precursor for the synthesis of other compounds that contain a methyl group.
The chemical formula of Acetic-1-13C-2-bromoacid-methylester is C4H6Br13COOCH3, and its molecular weight is 197.99 g/mol. The compound is a colorless liquid that is soluble in organic solvents such as chloroform, methanol, and ethanol.
In terms of its chemical properties, Acetic-1-13C-2-bromoacid-methylester is a strong electrophile due to the presence of the bromine atom, which makes it reactive towards nucleophiles such as amines, alcohols, and thiols. The compound can also undergo hydrolysis under acidic or basic conditions, which makes it a useful precursor for the synthesis of carboxylic acids.
Get an Instant Quote
This page contains information about Acetic-1-13C-2-bromoacid-methylester. You can buy Acetic-1-13C-2-bromoacid-methylester from Clearsynth at best competitive price with assured price guarantee. Clearsynth offers best quality Acetic-1-13C-2-bromoacid-methylester