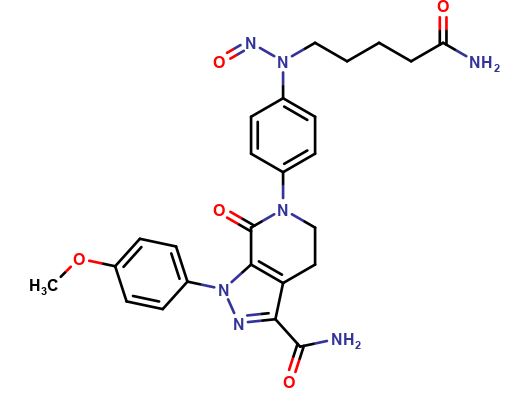
Apixaban Nitrosoamide impurity
Also known as: Apixaban Nitrosamine Impurities or nitrosamine impurities of Apixaban| Product Name | Apixaban Nitrosoamide impurity |
|---|---|
| Alternate Names | Apixaban Impurities, Impurities of Apixaban |
| CAT No. | CS-O-45383 |
| CAS No. | Not Available |
| Category | Impurities |
| Stock | Enquire |
| Mol. Wt. | 505.53 g/mol |
| Mol. For. | C25H27N7O5 |
| Hazardous | This is not a Hazardous Compound |
| COA | View Sample COA |
| MSDS | View Sample MSDS |
| Parent API | Apixaban |
| Controlled | No |
| Shipping | Free for purchase above 1000$ |
| Delivery | In-Stock, products will be dispatched within 24 hours via FedEx for USA, Europe, and other countries. |
| Return | Returns/replacement accepted if you are not satisfied with the quality of the product, (please send us an email with the reason/issues which are facing, within 15 days, after receipt of the product). |
| Ordering | Place your order online or by email sales@clearsynth.com |
Apixaban is a commonly used medication that is used to reduce the risk of stroke and blood clots in individuals with certain medical conditions. However, during the manufacturing process, impurities can be formed and one such impurity is Apixaban Nitrosoamide.
Apixaban Nitrosoamide is a type of organic compound that is formed during the production of Apixaban. It is important to note that this impurity can have harmful effects on the human body if consumed in large quantities. Therefore, it is important to closely monitor the levels of Apixaban Nitrosoamide in Apixaban formulations and ensure that they are within acceptable limits.
Chemically, Apixaban Nitrosoamide is a nitrosoamide derivative of Apixaban. It has a molecular formula of C23H20ClN7O3 and a molecular weight of 477.91 g/mol. This impurity is known to be a potential carcinogen and can cause damage to the DNA of cells in the body.
The acceptable limit of Apixaban Nitrosoamide impurity in Apixaban formulations is set by regulatory bodies such as the United States Pharmacopeia (USP). Manufacturers of Apixaban are required to test their products for the presence of this impurity and ensure that levels are within acceptable limits before they can be sold to consumers.
In conclusion, Apixaban Nitrosoamide is an impurity that can form during the production of Apixaban and can have harmful effects on the human body if consumed in large quantities. Therefore, it is important to closely monitor its levels in Apixaban formulations and ensure that they are within acceptable limits.
Get an Instant Quote
Related Compounds
Apixaban Amino Acid Impurity Hydrochloride | Desmethoxy Apixaban | Methyl 5-((4-(3-Carbamoyl-1-(4-methoxyphenyl)-7-oxo-4,5-dihydro-1H-pyrazolo[3,4-c]pyridin-6(7H)-yl)phenyl)amino)pentanoate | Apixaban Impurity 39 | Apixaban Impurity - (3/2 H2O) | Apixaban imp Dimer-2 | Apixaban Impurity 13 Potassium Salt | Apixaban Impurity 34 | N-Nitroso Apixaban Amino Acid Impurity | Apixaban Impurity 24 | Apixaban Impurity 27 | Apixaban dimer impurity | Apixaban methyl ester Dimer imp | Apixaban Impurity 17 | Apixaban Nitrosoacid impurity | Hydroxy O-Demethyl Apixaban | Apixaban impurity A | Apixaban Chloro Impurity | O-Desmethyl Apixaban | Apixaban Related Compound 5 | AXN-III | Apixaban impurity E | Apixaban Impurity 26 | Apixaban Related Compound H | Apixaban impurity D | Apixaban Hydroxy acid Impurity | Apixaban Related Compound G | Apixaban impurity B | Apixaban Impurity 12 | Apixaban Amino Acid Impurity | Apixaban Related Compound 1 | Apixaban PG Ester-II | N-(4-methoxyphenyl)nitrous hydrazide | Apixaban Impurity 32 | Ethyl (2E)-2-chloro-2-[2-(4-methoxyphenyl)hydrazinylidene]acetate | Apixaban Impurity 3 | Apixaban diacid impurity | Apixaban Impurity 28 | Apixaban Methyl ester Impurity | Apixaban Impurity 16 | Apixaban Impurity C | Apixaban Impurity 52 | Apixaban Ethoxy Diethylamide impurity | Apixaban Impurity - (2 H2O) | Apixaban Impurity 33 | O-Desmethyl Apixaban Sulfate | Apixaban carboxybutylaminophenyl | Apixaban PG Ester-I | Apixaban Impurity 44 | Ethyl-2-chloro-2-(2-(4-hydroxy phenyl)hydrazono)acetate | N-Nitroso 5-Chloropentanamide Apixaban | Apixaban Ester Impurity | ethyl (Z)-2-chloro-2-(2-(4-methoxyphenyl)hydrazineylidene)acetate | Apixaban Impurity 5 | Apixaban Impurity 38 | Apixaban Impurity 51 | Apixaban Impurity 84 | Apixaban Isopropyl Ester Impurity | Apixaban Impurity 14 | Apixaban Related Compound D | Apixaban Impurity 3 | Ethyl 2-chloro-2-[2-(2-methoxyphenyl)hydrazin-1-ylidene]acetate | Apixaban Impurity 43 | Apixaban Impurity 29 | Apixaban Impurity 40 |