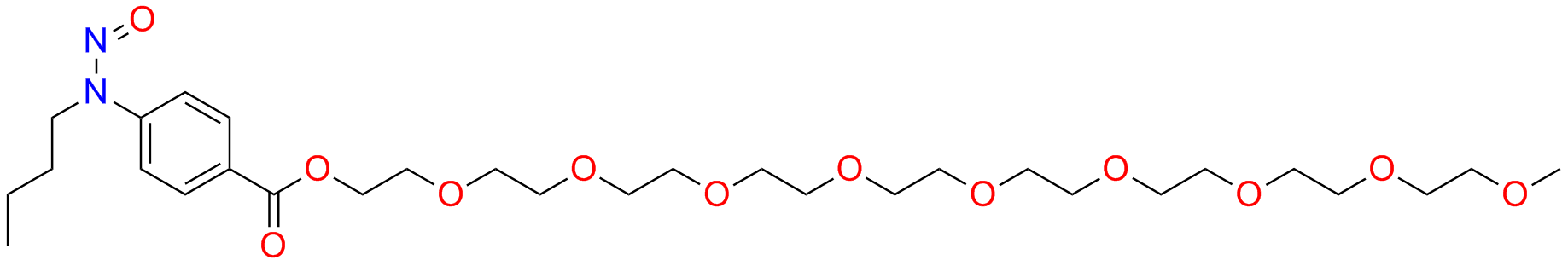
Benzonatate Nitroso Impurity
Also known as: Benzonatate Nitrosamine Impurities or nitrosamine impurities of Benzonatate| Product Name | Benzonatate Nitroso Impurity |
|---|---|
| Alternate Names | Benzonatate Impurities, Impurities of Benzonatate |
| CAT No. | CS-EO-02109 |
| CAS No. | Not Available |
| Category | Impurities |
| Stock | Enquire |
| Mol. Wt. | 632.7 g/mol |
| Mol. For. | C30H52N2O12 |
| Hazardous | This is not a Hazardous Compound |
| COA | View Sample COA |
| MSDS | View Sample MSDS |
| Parent API | Benzonatate |
| Controlled | No |
| Shipping | Free for purchase above 1000$ |
| Delivery | In-Stock, products will be dispatched within 24 hours via FedEx for USA, Europe, and other countries. |
| Return | Returns/replacement accepted if you are not satisfied with the quality of the product, (please send us an email with the reason/issues which are facing, within 15 days, after receipt of the product). |
| Ordering | Place your order online or by email sales@clearsynth.com |
Benzonatate is a medication used for the treatment of cough. It works by numbing the throat and lungs, reducing the urge to cough. However, like all medications, it can contain impurities. One such impurity is Benzonatate Nitroso Impurity.
Benzonatate Nitroso Impurity is a chemical compound that is formed during the manufacturing process of Benzonatate. It is a potential impurity that can be found in small amounts in Benzonatate capsules. The presence of this impurity can have adverse effects on the human body, and therefore it is essential to monitor its levels in the medication.
Chemically, Benzonatate Nitroso Impurity is a nitroso compound. It has a nitroso group (-NO) attached to a benzene ring. This impurity is formed when the nitrosation of an intermediate compound takes place during the manufacturing process of Benzonatate.
To ensure the safety of patients, regulatory authorities around the world have set limits on the levels of Benzonatate Nitroso Impurity that can be present in Benzonatate capsules. Manufacturers are required to test their products for this impurity and ensure that the levels are within the permissible limits before releasing them into the market.
In conclusion, Benzonatate Nitroso Impurity is a potential impurity found in Benzonatate capsules that requires monitoring to ensure patient safety. It is a nitroso compound that can be formed during the manufacturing process of Benzonatate. Regulatory authorities have set limits on its levels in Benzonatate capsules to ensure its safe usage.
Get an Instant Quote
This page contains information about Benzonatate Nitroso Impurity. You can buy Benzonatate Nitroso Impurity from Clearsynth at best competitive price with assured price guarantee. Clearsynth offers best quality Benzonatate Nitroso Impurity. This compound is also referred as Benzonatate nitrosamine impurities or nitrosamine impurities of Benzonatate