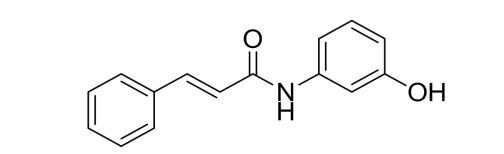
Brexpiprazole Impurity 19
| Product Name | Brexpiprazole Impurity 19 |
|---|---|
| Alternate Names | Brexpiprazole Impurities, Impurities of Brexpiprazole |
| CAT No. | CS-P-07261 |
| CAS No. | 616227-75-9 |
| Category | Impurities |
| Stock | IN-Stock |
| Mol. Wt. | Not Available |
| Mol. For. | Not Available |
| Hazardous | This is not a Hazardous Compound |
| COA | View Sample COA |
| MSDS | View Sample MSDS |
| Parent API | Brexpiprazole |
| Therapeutic | Anti anxiety and Anti Psychotic |
| Controlled | No |
| Shipping | Free for purchase above 1000$ |
| Delivery | In-Stock, products will be dispatched within 24 hours via FedEx for USA, Europe, and other countries. |
| Return | Returns/replacement accepted if you are not satisfied with the quality of the product, (please send us an email with the reason/issues which are facing, within 15 days, after receipt of the product). |
| Ordering | Place your order online or by email sales@clearsynth.com |
Brexpiprazole Impurity 19 is a chemical compound that is used for research and development purposes. It is a byproduct of the synthesis of brexpiprazole, a medication used to treat schizophrenia and depression. Brexpiprazole Impurity 19 is one of the many impurities that can be formed during the manufacturing process of brexpiprazole, and it is important to determine its presence and quantity to ensure the purity and safety of the final product.
Chemically, Brexpiprazole Impurity 19 is a complex organic compound with a molecular formula and structure that differ from those of brexpiprazole. It has a molecular weight of 441.51 g/mol and a melting point of approximately 154-156°C. The compound is sparingly soluble in water and soluble in organic solvents such as methanol, ethanol, and acetonitrile.
In research and development, Brexpiprazole Impurity 19 can be used as a reference standard or analytical marker for the identification and quantification of impurities in brexpiprazole samples. It can also be used to study the chemical properties and behavior of impurities in the manufacturing process of brexpiprazole, which can help improve the quality and efficiency of the process.
Overall, Brexpiprazole Impurity 19 is an important chemical compound in the field of pharmaceuticals and research, and its usage and chemical information are essential for ensuring the safety and efficacy of brexpiprazole and related medications.
Get an Instant Quote
Related Compounds
Brexpiprazole Impurity 7 | Brexpiprazole Dequinolinonyl | Brexpiprazole citrate | Brexpiprazole Impurity 10 | Brexpiprazole Impurity 13 | Brexpiprazole Impurity 5 | Brexpiprazole Impurity 2 | Brexpiprazole Impurity 31 | Brexpiprazole BBO Dimer Impurity | Brexpiprazole Impurity 15 | N-Nitroso Brexpiprazole piperazine intermediate | Brexpiprazole Impurity 14 | Brexpiprazole Impurity 6 | Brexpiprazole Impurity 41 | Brexpiprazole dihydrochloride | Brexpiprazole Impurity 22 | Brexpiprazole Impurity 30 | N-(benzo[b]thiophen-4-yl)-N-(2-(N-(4-((2-oxo-1,2-dihydroquinolin-7-yl)oxy)butyl)formamido)ethyl)form | Brexpiprazole Impurity 24 | Brexpiprazole Impurity 52 | N-[4-((2-Oxo-1,2-dihydroquinolin-7-yl)oxy)butyl]Brexpiprazole | Brexpiprazole Impurity 33 | Brexpiprazole Impurity 16 | Brexpiprazole Impurity 3 (Brexpiprazole N-Oxide) | Brexpiprazole Impurity 17 | Brexpiprazole Impurity 5 HCl salt | Brexpiprazole Impurity 11 | Brexpiprazole Impurity 12 | Brexpiprazole Impurity 27 | N-Nitroso Brexpiprazole Impurity 23 | Brexpiprazole Impurity 21 | Brexpiprazole Impurity 24 | Brexpiprazole Impurity 4 | Brexpiprazole Impurity 35 | Brexpiprazole Impurity 8 |