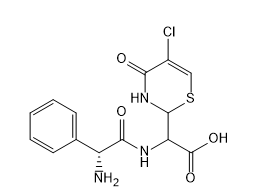
Cefaclor EP Iimpurity E
| Product Name | Cefaclor EP Iimpurity E |
|---|---|
| Alternate Names | Cefaclor Impurities, Impurities of Cefaclor |
| CAT No. | CS-P-03616 |
| CAS No. | 188915-50-6 |
| Category | Impurities |
| Stock | IN-Stock |
| Mol. Wt. | 355.80 g/mol |
| Mol. For. | C14H14ClN3O4S |
| Hazardous | This is a Hazardous Compound |
| COA | View Sample COA |
| MSDS | View Sample MSDS |
| Parent API | Cefaclor |
| Therapeutic | Antibiotics |
| Controlled | No |
| Shipping | Free for purchase above 1000$ |
| Delivery | In-Stock, products will be dispatched within 24 hours via FedEx for USA, Europe, and other countries. |
| Return | Returns/replacement accepted if you are not satisfied with the quality of the product, (please send us an email with the reason/issues which are facing, within 15 days, after receipt of the product). |
| Ordering | Place your order online or by email sales@clearsynth.com |
Cefaclor EP Impurity E is a potential impurity found in the manufacturing process of Cefaclor, a commonly used antibiotic medication. Cefaclor EP Impurity E is also known as 2-Chloro-6-(2,2,2-trifluoroethoxy)benzoic acid.
This impurity may be present in Cefaclor samples in small quantities and may not have any significant effect on the overall efficacy of the medication. However, there is a limit to the amount of impurity that is allowed in Cefaclor, and it must be monitored and controlled to ensure the safety and effectiveness of the medication.
Cefaclor is a second-generation cephalosporin antibiotic used to treat bacterial infections such as pneumonia, bronchitis, and ear infections. It works by inhibiting the growth and production of bacteria, ultimately leading to their elimination from the body.
As with any medication, it is important to follow the prescribed dosage and duration of treatment to ensure maximum effectiveness and minimize the risk of side effects. Patients should also inform their healthcare provider of any allergies or previous adverse reactions to antibiotics before starting treatment with Cefaclor.
In conclusion, Cefaclor EP Impurity E is a potential impurity found in Cefaclor samples that must be monitored and controlled to ensure the safety and effectiveness of the medication. Cefaclor is a commonly used antibiotic medication used to treat bacterial infections and should be taken as prescribed by a healthcare provider.
Get an Instant Quote
Related Compounds
Cefaclor EP Impurity F | Br-Analog Cefaclor | Cefaclor Impurity 1 | Cefaclor EP Impurity-B | Cefaclor EP Impurity C | Benzyl amine analog cefaclor | Cefaclor Impurity 5 | Cefaclor Impurity 4 | Cefaclor EP Impurity G | Cefaclor Impurity 8 (Cefaclor dimer) | Cefaclor EP Impurity H |