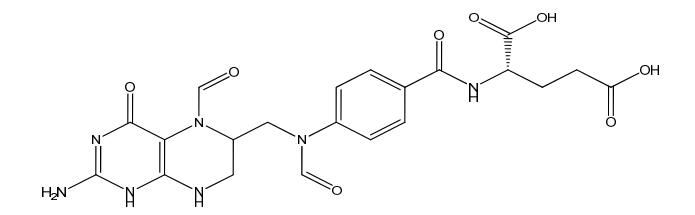
Folinic Acid Impurity B
| Product Name | Folinic Acid Impurity B |
|---|---|
| Alternate Names | Folinic Acid Impurities, Impurities of Folinic Acid |
| CAT No. | CS-P-07900 |
| CAS No. | 98814-60-9 |
| Category | Impurities |
| Stock | IN-Stock |
| Mol. Wt. | 501.5 g/mol |
| Mol. For. | C21H23N7O8 |
| Hazardous | This is a Hazardous Compound |
| COA | View Sample COA |
| MSDS | View Sample MSDS |
| Parent API | Folinic Acid |
| Purity | 95% |
| Therapeutic | Anti-Cancer / Oncology |
| Smileys | O=CN(C(CN1)CN(C2=CC=C(C(N[C@H](C(O)=O)CCC(O)=O)=O)C=C2)C=O)C3=C1NC(N)=NC3=O |
| Controlled | No |
| Shipping | Free for purchase above 1000$ |
| Delivery | In-Stock, products will be dispatched within 24 hours via FedEx for USA, Europe, and other countries. |
| Return | Returns/replacement accepted if you are not satisfied with the quality of the product, (please send us an email with the reason/issues which are facing, within 15 days, after receipt of the product). |
| Ordering | Place your order online or by email sales@clearsynth.com |
Folinic Acid Impurity B is a chemical compound that is often used in the manufacturing of pharmaceutical products. It has a molecular formula of C20H22N8O7 and a molecular weight of 498.44 g/mol. It is an impurity that is typically present during the synthesis of Folinic Acid, a medicinal compound that is used to treat certain types of anemia, as well as to reduce the toxic effects of chemotherapy.
Folinic Acid Impurity B is considered to be a minor impurity, and its presence typically does not affect the efficacy of the final product. However, it is important to monitor the levels of this impurity during the manufacturing process, as excessive amounts can potentially affect the quality and safety of the product.
In terms of usage, Folinic Acid Impurity B is typically removed from the final product through various purification processes, such as chromatography or crystallization. It is important to ensure that the final product meets the required specifications for purity and quality, as set forth by regulatory agencies such as the United States Pharmacopeia (USP) or the European Pharmacopoeia (EP).
Overall, Folinic Acid Impurity B is an important impurity to monitor and control during the manufacturing of Folinic Acid, as it can potentially affect the safety and efficacy of the final product. Through careful monitoring and purification processes, manufacturers can ensure that their products meet the required standards of quality and safety.
Get an Instant Quote
Related Compounds
Folinic Acid Impurity G | Folinic Acid Impurity H (Levofolinic Acid) | Folinic Acid Impurity B Acetate salt | Folinic Acid Impurity 5 (4-Aminobenzoyl D-Glutamic Acid) | Folinic Acid Impurity F | N-Nitroso Folinic acid (Leucovorin) | Folinic Acid Impurity E |