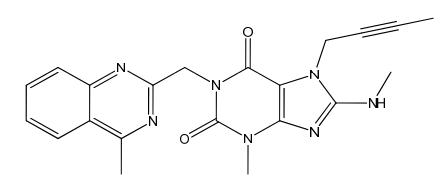
Linagliptin Impurity 49
| Product Name | Linagliptin Impurity 49 |
|---|---|
| Alternate Names | Linagliptin Impurities, Impurities of Linagliptin |
| CAT No. | CS-EO-01850 |
| CAS No. | 2468656-63-3 |
| Category | Impurities |
| Stock | IN-Stock |
| Mol. Wt. | 403.44 g/mol |
| Mol. For. | C₂₁H₂₁N₇O₂ |
| Hazardous | This is a Hazardous Compound |
| COA | View Sample COA |
| MSDS | View Sample MSDS |
| Parent API | Linagliptin |
| Controlled | No |
| Shipping | Free for purchase above 1000$ |
| Delivery | In-Stock, products will be dispatched within 24 hours via FedEx for USA, Europe, and other countries. |
| Return | Returns/replacement accepted if you are not satisfied with the quality of the product, (please send us an email with the reason/issues which are facing, within 15 days, after receipt of the product). |
| Ordering | Place your order online or by email sales@clearsynth.com |
Linagliptin Impurity 49 is a chemical compound that is commonly used in the pharmaceutical industry as a reference standard during the development and manufacturing of linagliptin-based drugs. It is also used as an analytical tool to detect and quantify impurities and other related substances in linagliptin drug substances and drug products.
Chemically, Linagliptin Impurity 49 is a derivative of pyrazole, which is a five-membered heterocyclic compound containing two nitrogen atoms in the ring. It is a white to off-white powder with a molecular weight of 222.23 g/mol.
The usage of Linagliptin Impurity 49 is strictly regulated and controlled under the guidelines of various regulatory authorities such as the US Food and Drug Administration (FDA) and the European Medicines Agency (EMA). These guidelines ensure the quality, safety, and efficacy of the final linagliptin product.
Linagliptin Impurity 49 is typically used in small quantities (parts per million) during the development, validation, and routine analysis of linagliptin drug substances and drug products. Its presence in the final product is closely monitored to ensure compliance with regulatory standards and to ensure the product's safety and efficacy.
In conclusion, Linagliptin Impurity 49 is an essential reference standard used in the pharmaceutical industry for the development, validation, and analysis of linagliptin-based drugs. Its usage is strictly regulated to ensure the quality, safety, and efficacy of the final product.
Get an Instant Quote
Related Compounds
N-Formyl Linagliptin | Linagliptin Impurity 2 | Linagliptin Impurity 1 | N-Hydroxy Linagliptin | Linagliptin impurity F | Linagliptin impurity K | Linagliptin Impurity Q | Linagliptin N-Acetyl Impurity | Linagliptin Impurity 4 | Linagliptin Impurity G | Linagliptin Impurity D | Linagliptin Dimer Impurity 4 | Linagliptin Impurity 9 | Linagliptin Impurity 29 | Linagliptin-d9 | Linagliptin Ethyldimer | Linagliptin impurity S | Linagliptin tert-butyl carbamate | Linagliptin Dimer Impurity | N-Boc Linagliptin | Linagliptin Impurity 37 | N-(6-Amino-1,2,3,4-tetrahydro-1-methyl-2,4-dioxo-5-pyrimidinyl)formamide | Linagliptin Diene Impurity | Linagliptin Hydroxy Impurity | Linagliptin N-Amino Acyl Impurity | Linagliptin Impurity 51 | Linagliptin impurity X | Linagliptin impurity P | Linagliptin Impurity 5 | Desmethyl N-nitroso Linagliptin | Linagliptin Impurity W | Linagliptin Impurity 42 | Linagliptin impurity H | N-Phthalimide-linagliptin | Linagliptin Impurity 76 TrisHCl salt | Linagliptin impurity B | Linagliptin impurity C | Linagliptin impurity 14 | Linagliptin Methyldimer | Linagliptin Impurity 3 | Linagliptin Dimer Impurity | Linagliptin Impurity 40 | Linagliptin Impurity 41 | Linagliptin Impurity 38 | Hydroxy Methyl Linagliptin | Linagliptin Impurity 36 | Linagliptin impurity M | Linagliptin impurity R | Desbutyl N-nitroso Linagliptin Impurity 52 | Desbutyl N-nitroso Linagliptin impurity 16 | Linagliptin Glycosamine | Desalkyl N-nitroso Linagliptin | Linagliptin Dimer impurity (N-C-C-C Dimer) | Linagliptin Impurity II | Linagliptin Impurity 43 | Linagliptin Impurity 25 | Linagliptin Impurity 41 | Linagliptin Impurity 106 | Linagliptin Pentahydroxy Impurity | Linagliptin Impurity-VII | N-nitroso Linagliptin Impurity C | Linagliptin impurity V | Linagliptin Impurity 17 | Linagliptin impurity 115 | Linagliptin impurity Y | Linagliptin Impurity 30 | Linagliptin impurity J | Linagliptin Impurity N | Linagliptin impurity U | Linagliptin Impurity 75 HCl salt |