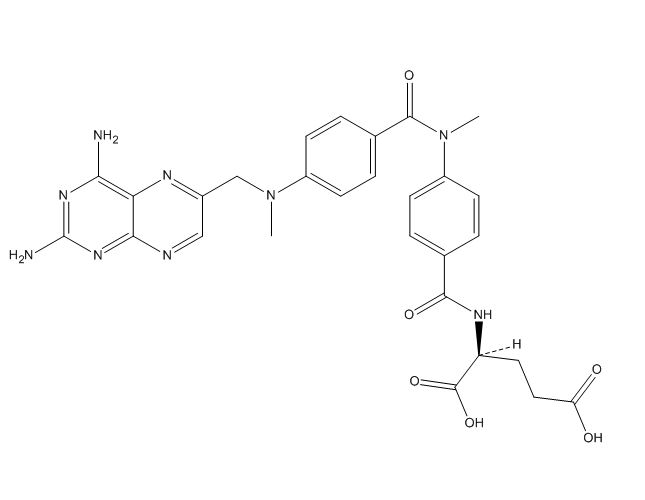
Methotrexate EP Impurity G
| Product Name | Methotrexate EP Impurity G |
|---|---|
| Alternate Names | Methotrexate Impurities, Impurities of Methotrexate |
| CAT No. | CS-EO-00339 |
| CAS No. | Not Available |
| Category | Impurities |
| Stock | IN-Stock |
| Mol. Wt. | 587.59 g/mol |
| Mol. For. | C₂₈H₂₉N₉O₆ |
| Hazardous | This is a Hazardous Compound |
| COA | View Sample COA |
| MSDS | View Sample MSDS |
| Parent API | Methotrexate |
| Controlled | No |
| Shipping | Free for purchase above 1000$ |
| Delivery | In-Stock, products will be dispatched within 24 hours via FedEx for USA, Europe, and other countries. |
| Return | Returns/replacement accepted if you are not satisfied with the quality of the product, (please send us an email with the reason/issues which are facing, within 15 days, after receipt of the product). |
| Ordering | Place your order online or by email sales@clearsynth.com |
Methotrexate EP Impurity G is a chemical compound that is used in the research and development of the drug Methotrexate, which is commonly used for the treatment of certain types of cancer, rheumatoid arthritis, and psoriasis. Methotrexate EP Impurity G is considered to be an impurity in the Methotrexate drug, and its presence can affect the purity and potency of the drug.
Chemically, Methotrexate EP Impurity G is a derivative of Methotrexate and has a molecular weight of 454.3 g/mol. Its chemical formula is C21H23N7O5 and it has a melting point of 273-275°C. The compound is a yellow to orange colored powder that is soluble in water and methanol.
Methotrexate EP Impurity G is primarily used as a reference standard for the development and validation of analytical methods for the detection and quantification of Methotrexate and its impurities. It is also used in the quality control of Methotrexate drug products to ensure their purity, potency, and safety.
In conclusion, Methotrexate EP Impurity G is an important chemical compound in the research and development of Methotrexate. Its usage and chemical information play a crucial role in the quality control and safety of Methotrexate drug products, ensuring that patients receive the most effective treatment possible.
Get an Instant Quote
Related Compounds
Methotrexate EP Impurity E | N-Nitroso Methotrexate EP Impurity-B | N-nitroso Methotrexate Ethylester impurity | N, N-Dinitroso Methotrexate EP Impurity L | Methotrexate EP Impurity F | Methotrexate EP Impurity A | N-Nitroso Methotrexate EP Impurity-L (1) | Methotrexate diethyl ester | N-Dinitroso Methotrexate Impurity 2 | R-Methotrexate di sodium | Methotrexate EP Impurity B Disodium salt | Methotrexate - Impurity E (HCl Salt) | Methotrexate Nitroso Impurity 1 | Methotrexate EP Impurity B (MonoHydrate) | Methotrexate EP Impurity L | Methotrexate EP Impurity C | Methotrexate EP Impurity D | N-Desmethyl Methotrexate dinitroso impurity | Methotrexate Dimethylamide | Methotrexate EP Impurity I | Methotrexate Nitroso Impurity 1 | Methotrexate EP Impurity J | Methotrexate EP Impurity H | Methotrexate EP Impurity B | N-nitroso Methotrexate Impurity 2 | N-Nitroso Methotrexate EP Impurity-L (2) |