Piracetam EP Impurity B
| Product Name | Piracetam EP Impurity B |
|---|---|
| Alternate Names | Piracetam Impurities, Impurities of Piracetam |
| CAT No. | CS-O-01919 |
| CAS No. | 59776-88-4 |
| Category | Impurities |
| Stock | IN-Stock |
| Mol. Wt. | 157.17 g/mol |
| Mol. For. | C₇H₁₁NO₃ |
| Hazardous | This is not a Hazardous Compound |
| COA | View Sample COA |
| MSDS | View Sample MSDS |
| Parent API | Piracetam |
| Purity | 96.25 |
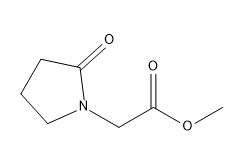
| Smileys | O=C(OC)CN(CCC1)C1=O |
| Canonical Smiles | COC(=O)CN1CCCC1=O |
| InchIKey | HPDZYDTXCOFUOY-UHFFFAOYSA-N |
| Inchl | InChI=1S/C7H11NO3/c1-11-7(10)5-8-4-2-3-6(8)9/h2-5H2,1H3 |
| IUPAC | methyl 2-(2-oxopyrrolidin-1-yl)acetate |
| Controlled | No |
| Shipping | Free for purchase above 1000$ |
| Delivery | In-Stock, products will be dispatched within 24 hours via FedEx for USA, Europe, and other countries. |
| Return | Returns/replacement accepted if you are not satisfied with the quality of the product, (please send us an email with the reason/issues which are facing, within 15 days, after receipt of the product). |
| Ordering | Place your order online or by email sales@clearsynth.com |
Piracetam EP Impurity B is a chemical compound that is commonly used in the pharmaceutical industry as a reference standard for the identification and quantification of impurities in the drug, Piracetam. Piracetam is a nootropic drug that is used to enhance cognitive function and memory in patients with dementia, Alzheimer's disease, and other neurological disorders. The impurities present in Piracetam can affect the efficacy and safety of the drug, hence the need for reference standards.
Piracetam EP Impurity B is a white to off-white powder that has a molecular weight of 169.2 g/mol. Its chemical formula is C9H9NO3S, and it has a melting point range of 118-122°C. This compound is classified as a thiazolidine-2,4-dione derivative and is structurally related to Piracetam.
In terms of usage, Piracetam EP Impurity B is primarily used as a reference standard for the identification and quantification of impurities in Piracetam. It is also used in the development and validation of analytical methods for impurity profiling of Piracetam. This compound is typically supplied in a certified reference material (CRM) form, which is a quality-controlled material that is used to validate analytical methods.
Overall, Piracetam EP Impurity B plays a critical role in ensuring the quality, safety, and efficacy of Piracetam as a pharmaceutical product. It serves as a valuable tool for pharmaceutical companies and regulatory agencies in the quality control and assurance of Piracetam.
Get an Instant Quote
This page contains information about Piracetam EP Impurity B. You can buy Piracetam EP Impurity B from Clearsynth at best competitive price with assured price guarantee. Clearsynth offers best quality Piracetam EP Impurity B