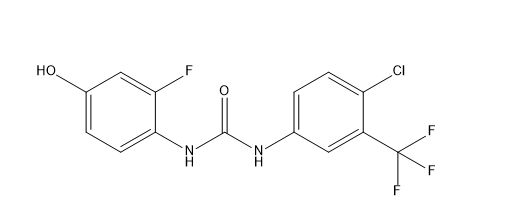
Regorafenib Urea Impurity
| Product Name | Regorafenib Urea Impurity |
|---|---|
| Alternate Names | Regorafenib Impurities, Impurities of Regorafenib |
| CAT No. | CS-O-14434 |
| CAS No. | 1333390-56-9 |
| Category | Impurities |
| Stock | IN-Stock |
| Mol. Wt. | 348.68 g/mol |
| Mol. For. | C₁₄H₉ClF₄N₂O₂ |
| Hazardous | This is a Hazardous Compound |
| COA | View Sample COA |
| MSDS | View Sample MSDS |
| Parent API | Regorafenib |
| Purity | >98% |
| Therapeutic | Anti-Cancer / Oncology |
| Smileys | FC(F)(C1=CC(NC(NC(C=CC(O)=C2)=C2F)=O)=CC=C1Cl)F |
| Controlled | No |
| Shipping | Free for purchase above 1000$ |
| Delivery | In-Stock, products will be dispatched within 24 hours via FedEx for USA, Europe, and other countries. |
| Return | Returns/replacement accepted if you are not satisfied with the quality of the product, (please send us an email with the reason/issues which are facing, within 15 days, after receipt of the product). |
| Ordering | Place your order online or by email sales@clearsynth.com |
Regorafenib is a multi-kinase inhibitor used in the treatment of metastatic colorectal cancer and gastrointestinal stromal tumors. It works by inhibiting angiogenesis and tumor growth. Regorafenib is a relatively new drug and is still under investigation for its efficacy in treating other types of cancer.
Regorafenib Urea Impurity is an impurity that can be found in Regorafenib. It is a urea derivative and is formed during the manufacturing process of Regorafenib. Urea impurities are common in pharmaceuticals and can be formed due to various factors such as reaction conditions, impurities in starting materials, and degradation of the drug.
The presence of Regorafenib Urea Impurity in Regorafenib does not affect its therapeutic effect. However, it is important to control the impurity levels as they may affect the drug’s stability and shelf life. The acceptable limit for Regorafenib Urea Impurity is set by regulatory authorities such as the US FDA.
Chemically, Regorafenib Urea Impurity is a white solid that is sparingly soluble in water. Its molecular formula is C22H23ClFN5O3 and its molecular weight is 457.90 g/mol.
In conclusion, Regorafenib Urea Impurity is a urea derivative that can be found in Regorafenib. Although it does not affect the drug’s therapeutic effect, it is important to control its levels to ensure the drug’s stability and shelf life.
Get an Instant Quote
Related Compounds
Regorafenib Impurity 1 | N-Desmethyl N-Hydroxymethyl Regorafenib | Regorafenib Impurity 4 | Regorafenib acid Impurity | Regorafenib Impurity 3 | Regorafenib Impurity 2 | Regorafenib Ethyl Carbamate Impurity | Regorafenib Dipyrimidine Impurity | Regorafenib EP Impurity D | AFP-Picolin Methylamide Impurity |