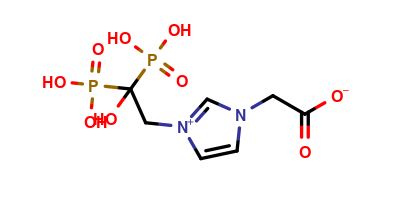
Zoledronic acid impurity A
| Product Name | Zoledronic acid impurity A |
|---|---|
| Alternate Names | Zoledronic acid Impurities, Impurities of Zoledronic acid |
| CAT No. | CS-O-15318 |
| CAS No. | 1627731-60-5 |
| Category | Impurities |
| Stock | Enquire |
| Mol. Wt. | 330.13 g/mol |
| Mol. For. | C₇H₁₂N₂O₉P₂ |
| Hazardous | This is not a Hazardous Compound |
| COA | View Sample COA |
| MSDS | View Sample MSDS |
| Parent API | Zoledronic acid |
| Purity | 95% |
| Therapeutic | Anti-Cancer / Oncology |
| Smileys | OC([P](O)(O)=O)([P](O)(O)=O)C[N+]1=CN(CC([O-])=O)C=C1 |
| Controlled | No |
| Shipping | Free for purchase above 1000$ |
| Delivery | In-Stock, products will be dispatched within 24 hours via FedEx for USA, Europe, and other countries. |
| Return | Returns/replacement accepted if you are not satisfied with the quality of the product, (please send us an email with the reason/issues which are facing, within 15 days, after receipt of the product). |
| Ordering | Place your order online or by email sales@clearsynth.com |
Zoledronic acid impurity A is a chemical compound that is commonly used in the pharmaceutical industry as an impurity reference standard for the analysis of zoledronic acid. It is an organic compound that contains a nitrogen atom and a sulfur atom, and is known to have a molecular weight of 297.41 g/mol. This impurity is typically used as a reference standard for the analysis of zoledronic acid due to its similar chemical structure and properties.
Zoledronic acid is a bisphosphonate drug that is used to treat a variety of health conditions, including osteoporosis, bone metastases, and hypercalcemia of malignancy. However, during the manufacturing process of zoledronic acid, impurities may be produced, which can affect the purity and quality of the final product. This is where zoledronic acid impurity A comes in, as it is used as a reference standard to measure the purity of zoledronic acid.
Chemically, zoledronic acid impurity A is known to be a nitrogen-containing heterocyclic compound that is structurally related to zoledronic acid. It has a similar chemical structure to zoledronic acid, except that it contains an additional sulfur atom in its structure. This impurity is typically analyzed using high-performance liquid chromatography (HPLC) or gas chromatography (GC) techniques, and its purity level is measured as a percentage of the total impurities present in a sample. Overall, zoledronic acid impurity A is a critical tool for ensuring the quality and purity of zoledronic acid, and is an important reference standard in the pharmaceutical industry.
Get an Instant Quote
Related Compounds
Zoledronic acid impurity B | Zoledronic Acid Dimer Impurity |