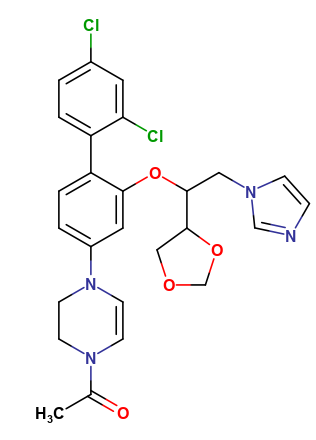
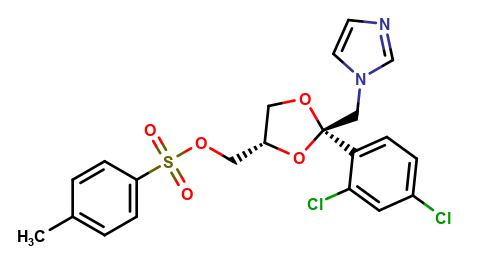
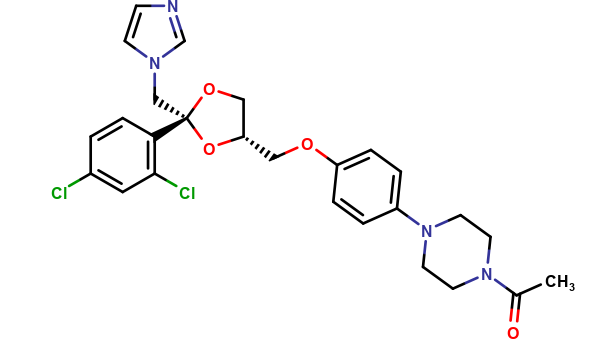
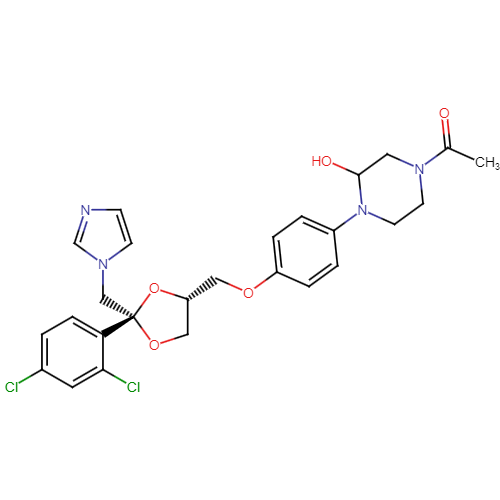
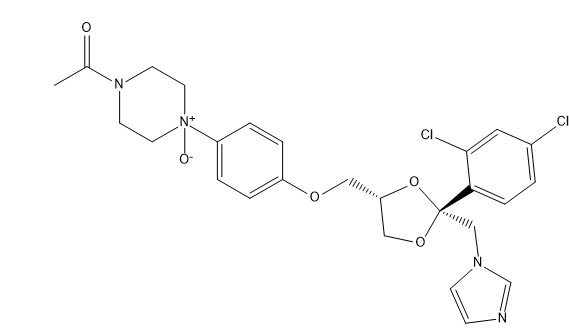
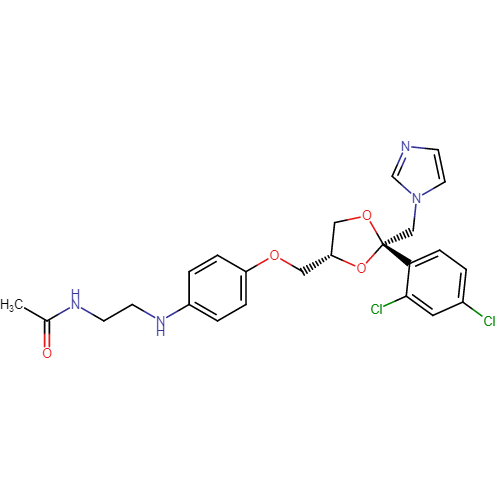
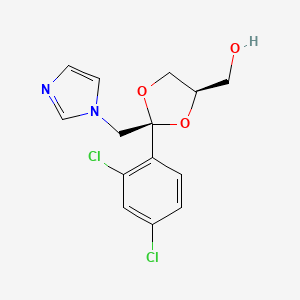
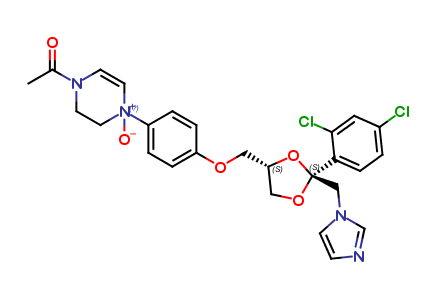
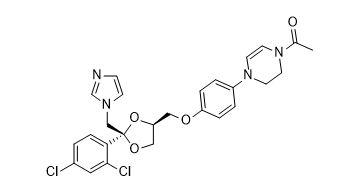
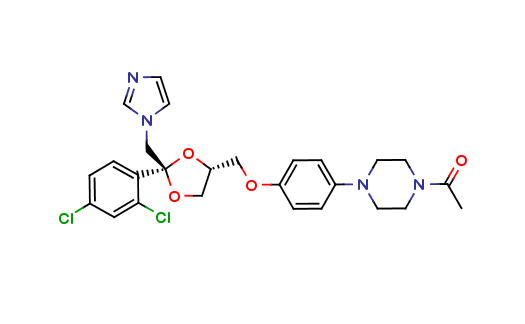
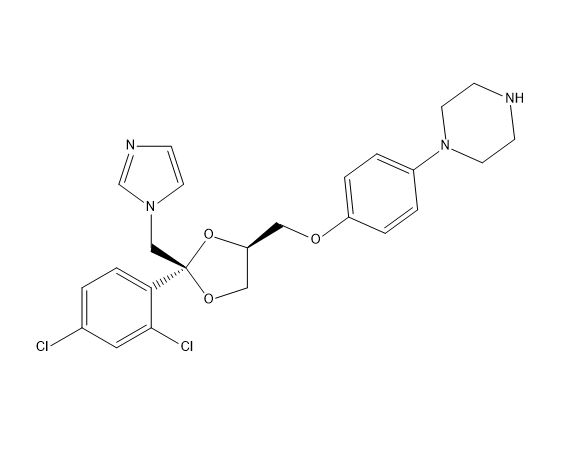
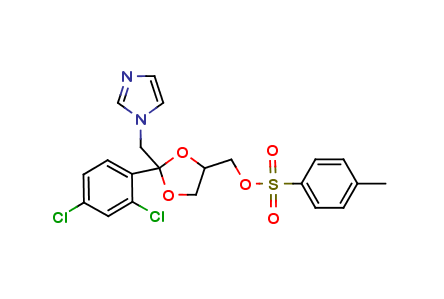
Ketoconazole Impurities and its Related Products
Ketoconazole Impurities refers to the unwanted substances or byproducts that may be present during the synthesis or manufacturing process of Ketoconazole. These impurities can affect the purity, quality, and effectiveness of the final product. It is essential to identify, quantify, and control these impurities to ensure that the end product is safe for consumption and meets the regulatory requirements. Some common impurities of Ketoconazole include related substances, degradation products, and residual solvents.