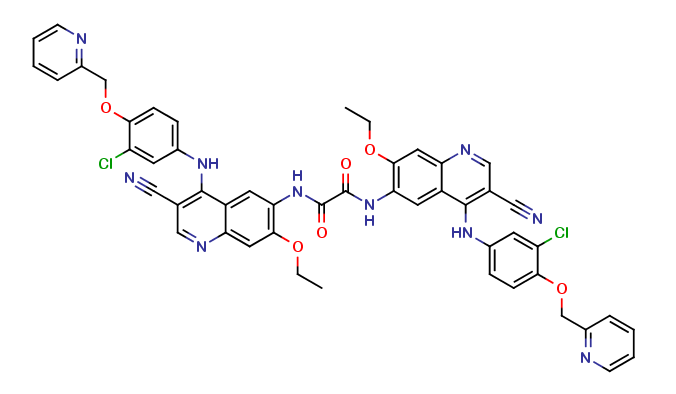
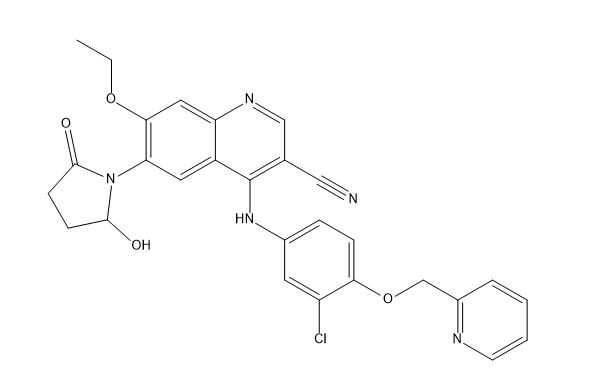
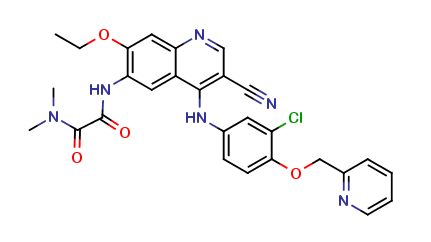
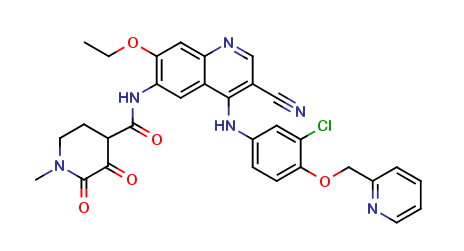
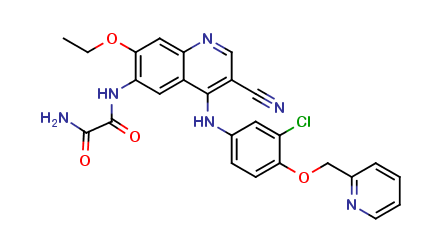
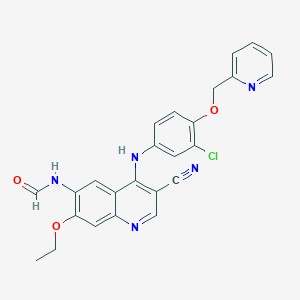
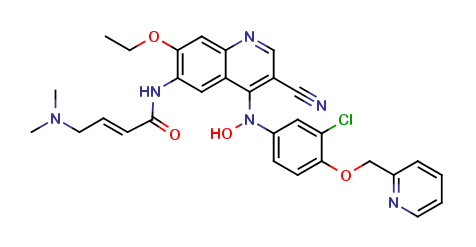
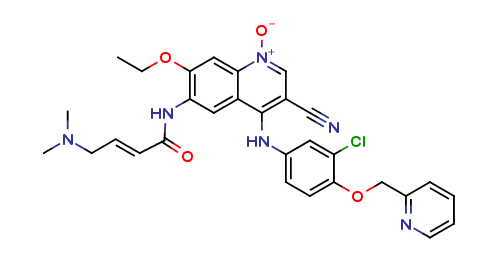
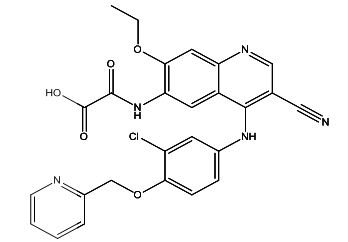
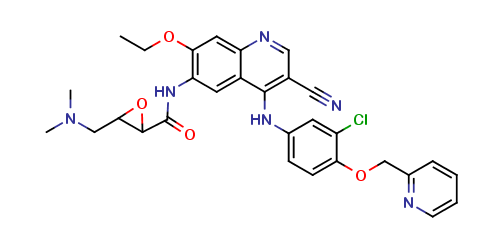
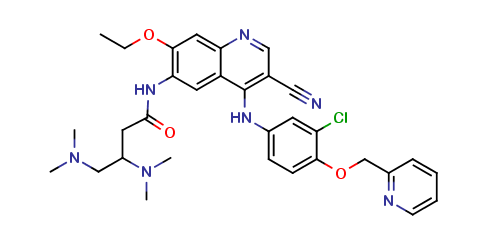
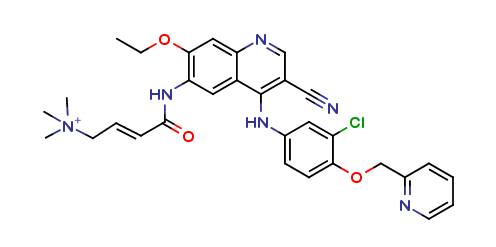
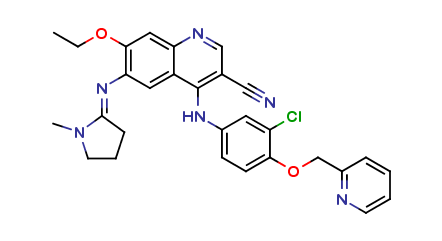
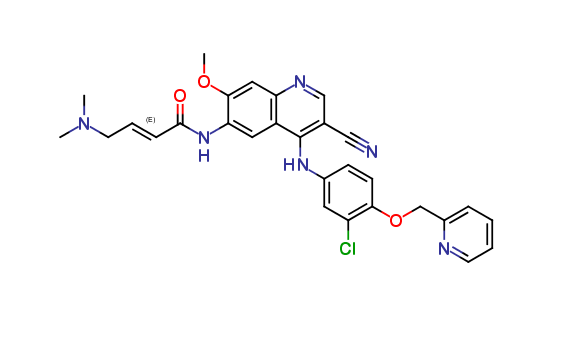
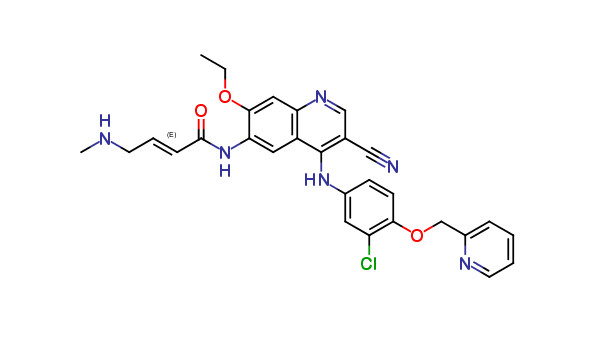
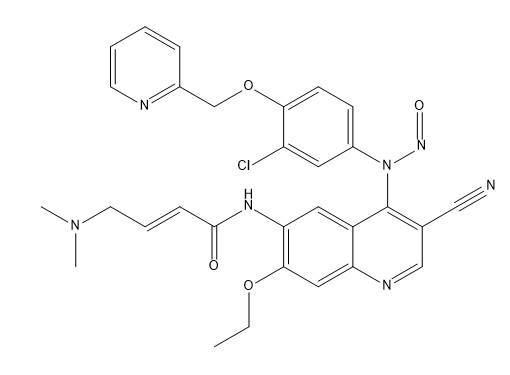
Neratinib Impurities and its Related Products
Neratinib is a potent irreversible inhibitor of the human epidermal growth factor receptor (HER) family of tyrosine kinase, used in the treatment of breast cancer. However, during the synthesis and manufacturing process of Neratinib, various impurities may be formed which can affect its safety, efficacy and shelf-life. These impurities need to be identified, characterized and controlled to ensure the quality and safety of the final drug product.