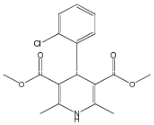
Amlodipine EP Impurity G
| Product Name | Amlodipine EP Impurity G |
|---|---|
| Alternate Names | Amlodipine Impurities, Impurities of Amlodipine |
| CAT No. | CS-O-30465 |
| CAS No. | 43067-01-2 |
| Category | Impurities |
| Stock | IN-Stock |
| Mol. Wt. | 335.78 g/mol |
| Mol. For. | C₁₇H₁₈ClNO₄ |
| Hazardous | This is a Hazardous Compound |
| COA | View Sample COA |
| MSDS | View Sample MSDS |
| Parent API | Amlodipine |
| Purity | 95% |
| Therapeutic | Anti-Hypertensives |
| Smileys | CC1=C(C(C(=C(N1)C)C(=O)OC)C2=CC=CC=C2Cl)C(=O)OC |
| Controlled | No |
| Shipping | Free for purchase above 1000$ |
| Delivery | In-Stock, products will be dispatched within 24 hours via FedEx for USA, Europe, and other countries. |
| Return | Returns/replacement accepted if you are not satisfied with the quality of the product, (please send us an email with the reason/issues which are facing, within 15 days, after receipt of the product). |
| Ordering | Place your order online or by email sales@clearsynth.com |
Amlodipine EP Impurity G is a chemical compound that is commonly used as a reference standard in the pharmaceutical industry. It is an impurity of Amlodipine, a medication used to treat high blood pressure and angina (chest pain). Amlodipine EP Impurity G is used to ensure the quality and purity of Amlodipine during its manufacturing process and to determine the presence of impurities in the final product.
Chemically, Amlodipine EP Impurity G is known as 2,6-dimethyl-4-(2-nitrophenyl)-1,4-dihydropyridine-3,5-dicarboxylic acid. It has a molecular weight of 345.34 g/mol and a melting point of 259-262°C. The compound is usually supplied as a white to off-white powder that is soluble in most organic solvents.
Amlodipine EP Impurity G is typically used in analytical chemistry as a reference standard in high-performance liquid chromatography (HPLC) and gas chromatography (GC) methods. It is also used in research and development to investigate the degradation products of Amlodipine and to develop new analytical methods for its quantification.
In summary, Amlodipine EP Impurity G is a useful reference standard in the pharmaceutical industry for the quality control of Amlodipine and for analytical chemistry purposes. Its chemical properties and analytical uses make it an important compound in the field of pharmaceutical research and development.
Get an Instant Quote
Related Compounds
Amlodipine N-Galactose | Amlodipine Aspartic Acid Impurity | Amlodipine orotate | Amlodipine Impurity 32 | Desmethyl Amlodipine | Di-acid Amlodipine | N-Fomyl Amlodipine | Atorvastatin Amlodipine Dimer | Amlodipine Nitroso Impurity | Deschloro Amlodipine | Amlodipine EP Impurity E | Amlodipine Impurity | Amlodipine EP Impurity A | Amlodipine Azido Impurity | Amlodipine EP Impurity C | Amlodipine aminoethyl | R-Amlodipine hemipentahydrate | Amlodipine EP Impurity D Oxalate Salt | Amlodipine Galactose Adduct | Atorvastatin-Amlodipine Adduct | Amlodipine Impurity 34 | Amlodipine Besylate Impurity D Besylate salt | Dehydro Amlodipine N-Oxide | Amlodipine Mannitol Adduct | Phthaloyl Amlodipine Dimethyl Ester | Amlodipine EP Impurity B | Amlodipine Mannitol Adduct Acetate Salt | Amlodipine N-Lactoside | Amlodipine EP Impurity D HCl | Amlodipine Related Compound A | Amlodipine Impurity F Maleate | N-Nitroso Amlodipine EP Impurity F | Amlodipine Impurity 38 | Amlodipine Lactose adduct | Amlodipine Impurity 61 | Amlodipin EP Impurity D | Amlodipine Impurity 9 | Amlodipine Impurity 19 | Amlodipine Impurity 8 | Amlodipine Mannitol Adduct HCl Salt | Amlodipine EP Impurity F | Amlodipine EP Impurity E Maleate | Amlodipine morpholine impurity | R-Amlodipine Besilate | Amlodipine Methyl Ester | Amlodipine EP Impurity H | Amlodipine For Peak Identification | Amlodipine Impurity 29 | Amlodipine Impurity 25 | Dimethoxy Impurity Deschloro Amlodipine | N-Nitroso Amlodipine | Hydroxyethyl phthalyl amlodipine |