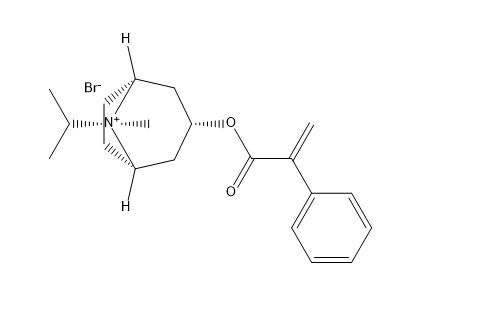
Ipratropium Bromide impurity F
| Product Name | Ipratropium Bromide impurity F |
|---|---|
| Alternate Names | Ipratropium Impurities, Impurities of Ipratropium |
| CAT No. | CS-O-15750 |
| CAS No. | 17812-46-3 |
| Category | Impurities |
| Stock | IN-Stock |
| Mol. Wt. | 394.35 g/mol |
| Mol. For. | C₂₀H₂₈BrNO₂ |
| Hazardous | This is a Hazardous Compound |
| COA | View Sample COA |
| MSDS | View Sample MSDS |
| Parent API | Ipratropium |
| Purity | 95% |
| Therapeutic | Anti-Asthma / COPD |
| Smileys | CC(C)[N+]1(C2CCC23C1CC(C3)OC(=O)C(=C)C4=CC=CC=C4)C.[Br-] |
| Controlled | No |
| Shipping | Free for purchase above 1000$ |
| Delivery | In-Stock, products will be dispatched within 24 hours via FedEx for USA, Europe, and other countries. |
| Return | Returns/replacement accepted if you are not satisfied with the quality of the product, (please send us an email with the reason/issues which are facing, within 15 days, after receipt of the product). |
| Ordering | Place your order online or by email sales@clearsynth.com |
Ipratropium Bromide impurity F is a chemical compound that is commonly used in the pharmaceutical industry. This impurity is a byproduct of the manufacturing process of Ipratropium Bromide, which is a medication used to treat respiratory conditions such as chronic obstructive pulmonary disease (COPD) and asthma.
The usage of Ipratropium Bromide impurity F is primarily in the pharmaceutical industry as a reference standard for quality control purposes. It is used to identify and quantify the impurity levels in Ipratropium Bromide formulations. This impurity can affect the efficacy and safety of the medication, so it is important to monitor its levels and ensure that they are within acceptable limits.
Chemically, Ipratropium Bromide impurity F is an organic compound with the molecular formula C20H28BrNO3. It is a white to off-white crystalline powder with a melting point of 135-138°C. This impurity is a quaternary ammonium derivative that contains a bromide ion. It is soluble in water and ethanol but insoluble in ether and chloroform.
In conclusion, Ipratropium Bromide impurity F is an important chemical compound in the pharmaceutical industry. Its usage as a reference standard for quality control purposes ensures the safety and efficacy of Ipratropium Bromide medications. The chemical information of this impurity is essential in identifying and quantifying its levels in formulations.
Get an Instant Quote
Related Compounds
Ipratropium EP Impurity B | Ipratropium Bromide Impurity 3 | Ipratropium-d7 Bromide | Ipratropium Bromide Impurity 2 | Des-Methoxy Ipratropium | Ipratropium EP Impurity D | Ipratropium EP Impurity E | Ipratropium EP Impurity C | Ipratropium Bromide Impurity A |