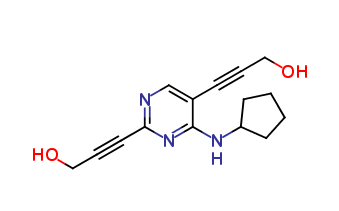
Ribociclib impurity-2
| Product Name | Ribociclib impurity-2 |
|---|---|
| Alternate Names | Ribociclib Impurities, Impurities of Ribociclib |
| CAT No. | CS-O-32727 |
| CAS No. | Not Available |
| Category | Impurities |
| Stock | IN-Stock |
| Mol. Wt. | 271.31 g/mol |
| Mol. For. | C15H17N3O2 |
| Hazardous | This is not a Hazardous Compound |
| COA | View Sample COA |
| MSDS | View Sample MSDS |
| Parent API | Ribociclib |
| Smileys | OCC#CC1=NC=C(C#CCO)C(NC2CCCC2)=N1 |
| Controlled | No |
| Shipping | Free for purchase above 1000$ |
| Delivery | In-Stock, products will be dispatched within 24 hours via FedEx for USA, Europe, and other countries. |
| Return | Returns/replacement accepted if you are not satisfied with the quality of the product, (please send us an email with the reason/issues which are facing, within 15 days, after receipt of the product). |
| Ordering | Place your order online or by email sales@clearsynth.com |
Ribociclib impurity-2 is a chemical compound that is used in the pharmaceutical industry as a reference standard for the detection and quantification of impurities in the active ingredient ribociclib. Ribociclib is a selective inhibitor of cyclin-dependent kinases 4 and 6 (CDK4/6) and is used in the treatment of hormone receptor-positive breast cancer. Ribociclib impurity-2 is a synthetic compound that is chemically related to ribociclib and has a similar structure. It is used as a reference standard in the quality control of ribociclib formulations and is also used in research and development of new drugs.
Chemically, ribociclib impurity-2 is a white to off-white solid that is soluble in organic solvents such as methanol, ethanol, and acetonitrile. Its molecular formula is C16H19N5O3 and its molecular weight is 329.36 g/mol. It has a melting point of 165-167°C and a purity of at least 98%.
The usage of ribociclib impurity-2 is important in ensuring the safety and efficacy of ribociclib formulations. Impurities in pharmaceutical products can affect their stability, bioavailability, and safety, and therefore it is necessary to monitor and control impurities at every stage of drug development and manufacturing. Ribociclib impurity-2 is a valuable tool in this process, as it enables accurate detection and quantification of impurities in ribociclib formulations.
Get an Instant Quote
Related Compounds
Ribociclib Impurity 29 | Ribociclib N-oxobutanoic acid | Ribociclib Mehyl Ester | Ribociclib Nitroso Impurity 1 | Ribociclib Chloro Hydroxymethyl Impurity | N-Trityl Ribociclib impurity | Ribociclib Succinate Amine impurity | Ribociclib Succinate Amino trityl impurity | Ribociclib Chloro Amide Impurity | Ribociclib impurity-7 | N-formyl Ribociclib | Ribociclib open ring formamide impurity | Ribociclib ACID IMPURITY | Ribociclib N-Oxide | Ribociclib Succinate Piperazine amide impurity | Ribociclib impurity-5 | Ribociclib Succinate Triphenyl isopropyl ether impurity | Ribociclib Impurity 12 | Ribociclib succinate | Ribociclib Impurity 13 | Ribociclib Nitroso Impurity 3 | Ribociclib impurity-9 | Ribociclib Nitroso Impurity 2 | Ribociclib impurity-10 |