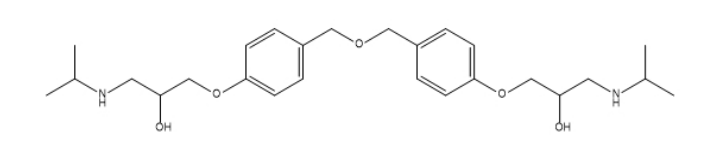
Bisoprolol EP Impurity D
| Product Name | Bisoprolol EP Impurity D |
|---|---|
| Alternate Names | Bisoprolol Impurities, Impurities of Bisoprolol |
| CAT No. | CS-O-07376 |
| CAS No. | 1225195-71-0 |
| Category | Impurities |
| Stock | IN-Stock |
| Mol. Wt. | 460.62 g/mol |
| Mol. For. | C₂₆H₄₀N₂O₅ |
| Hazardous | This is a Hazardous Compound |
| COA | View Sample COA |
| MSDS | View Sample MSDS |
| Parent API | Bisoprolol |
| Therapeutic | Anti-Hypertensives |
| Smileys | OC(CNC(C)C)COC1=CC=C(COCC2=CC=C(OCC(O)CNC(C)C)C=C2)C=C1 |
| Controlled | No |
| Shipping | Free for purchase above 1000$ |
| Delivery | In-Stock, products will be dispatched within 24 hours via FedEx for USA, Europe, and other countries. |
| Return | Returns/replacement accepted if you are not satisfied with the quality of the product, (please send us an email with the reason/issues which are facing, within 15 days, after receipt of the product). |
| Ordering | Place your order online or by email sales@clearsynth.com |
Bisoprolol is a beta-blocker medication used primarily for the treatment of high blood pressure and heart failure. Bisoprolol EP Impurity D is a chemical compound that is related to bisoprolol and is considered an impurity in the manufacturing process of bisoprolol.
Bisoprolol EP Impurity D is a white to off-white crystalline powder that is insoluble in water but soluble in organic solvents such as methanol and acetonitrile. It is typically present in very small quantities in commercial batches of bisoprolol, usually less than 0.05% by weight.
While the presence of Bisoprolol EP Impurity D is not desirable in commercial batches of bisoprolol, it is not considered to be harmful to patients who take the medication. The impurity is not pharmacologically active and has not been found to cause any adverse effects on the body.
Manufacturers of bisoprolol are required to monitor and control the levels of Bisoprolol EP Impurity D in their products to ensure that they meet regulatory standards. This involves using analytical techniques such as high-performance liquid chromatography (HPLC) to measure the impurity levels and adjust the manufacturing process as needed to minimize its presence.
In conclusion, Bisoprolol EP Impurity D is a minor impurity in the manufacturing process of bisoprolol that does not pose a threat to patient safety. Its levels are monitored and controlled by manufacturers to ensure regulatory compliance.
Get an Instant Quote
Related Compounds
Bisoprolol EP Impurity F | R-(+)-Bisoprolol Fumarate | Bisoprolol glycerol diether impurity | Bisoprolol EP Impurity F Butenedioic acid salt | Bisoprolol Benzylalcohol | Bisoprolol EP Impurity G fumarate salt | N-Formylbisoprolol | Bisoprolol EP Impurity A | Bisoprolol Impurity 11 (mixture of isomers) | Bisoprolol Related Compound D | Bisoprolol EP Impurity C | Bisoprolol N-Methyl Impurity | Bisoprolol EP Impurity M | Bisoprolol Phenol Impurity | N-Acetyl Bisoprolol | Bisoprolol EP Impurity L hydrochloride | Bisoprolol EP Impurity Q | Bisoprolol EP Impurity B | Bisoprolol EP Impurity D (TFA) | Bisoprolol O-Phosphate | Bisoprolol Alcohol Impurity | Bisoprolol Dimer | Bisoprolol benzyl alcohol fumarate | Bisoprolol Epoxide Impurity | Dehydro Bisoprolol Hemifumarate | Bisoprolol EP Impurity N | Bisoprolol Impurity 4 | Des O-isopropyl Bisoprolol | Bisoprolol Impurity 3 | Bisoprolol EP Impurity R | Bisoprolol EP Impurity T | Dehydro Bisoprolol | N-Desisopropyl-N-formyl Bisoprolol | Bisoprolol Impurity | Bisoprolol EP Impurity B Hemifumarate (Bisoprolol n-Propyl Derivative Hemifumarate) | Bisoprolol Carboxylic Acid Impurity | Bisoprolol Diene Impurity | Bisoprolol EP Impurity J | Bisoprolol Ester Impurity |