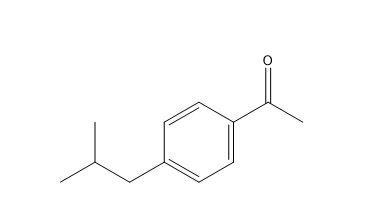
Ibuprofen USP Related Compound C
| Product Name | Ibuprofen USP Related Compound C |
|---|---|
| Alternate Names | Ibuprofen Impurities, Impurities of Ibuprofen |
| CAT No. | CS-O-11051 |
| CAS No. | 38861-78-8 |
| Category | Impurities |
| Stock | IN-Stock |
| Mol. Wt. | 176.25 g/mol |
| Mol. For. | C12H16O |
| Hazardous | This is a Hazardous Compound |
| COA | View Sample COA |
| MSDS | View Sample MSDS |
| Parent API | Ibuprofen |
| Purity | Not less than 98 % |
| Therapeutic | Anti-Asthma / COPD |
| Smileys | CC(C)CC1=CC=C(C=C1)C(=O)C |
| Controlled | No |
| Shipping | Free for purchase above 1000$ |
| Delivery | In-Stock, products will be dispatched within 24 hours via FedEx for USA, Europe, and other countries. |
| Return | Returns/replacement accepted if you are not satisfied with the quality of the product, (please send us an email with the reason/issues which are facing, within 15 days, after receipt of the product). |
| Ordering | Place your order online or by email sales@clearsynth.com |
Ibuprofen USP Related Compound C is a chemical substance that is commonly used in the pharmaceutical industry as a reference standard for the analysis of ibuprofen. Ibuprofen is a non-steroidal anti-inflammatory drug (NSAID) that is used to relieve pain, inflammation, and fever. Ibuprofen USP Related Compound C, also known as 2-(4-isobutylphenyl) propionic acid, is a closely related compound to ibuprofen that is used as a marker or impurity in the manufacturing process of ibuprofen.
Chemically, Ibuprofen USP Related Compound C is a white, crystalline substance with a molecular weight of 206.3 g/mol and a melting point of 61-63°C. It is soluble in ethanol, methanol, and dimethyl sulfoxide.
The usage of Ibuprofen USP Related Compound C in the pharmaceutical industry is mainly for quality control purposes. It is used as a reference standard to ensure the purity of ibuprofen in drug formulations. The presence of Ibuprofen USP Related Compound C in ibuprofen drug formulations is monitored to ensure that it does not exceed the acceptable limits, as it could potentially affect the efficacy and safety of the drug.
In conclusion, Ibuprofen USP Related Compound C is an important reference standard in the pharmaceutical industry for the analysis and quality control of ibuprofen. Its usage ensures that ibuprofen drug formulations are of the highest quality and meet the required standards for safety and efficacy.
Get an Instant Quote
Related Compounds
Ibuprofen Sorbitan Ester | Ibuprofen Impurity R | Ibuprofen Impurity D | Ibuprofen EP Impurity K | Tetraethyleneglycol Bisibuprofen Ester | Ibuprofen Methyl Ester | Ibuprofen Impurity 27 | Ibuprofen EP Impurity B | Ibuprofen EP Impurity I | Ibuprofen impurity N | Ibuprofen Alcohol | Ibuprofen Piconol | Ibuprofen impurity at RRT 1.76 | Ibuprofen 1,2,3-Propanetriol Esters (Mixture of Regio- and Stereoisomers) | Ibuprofen Impurity I | Ibuprofen EP Impurity A | Ibuprofen 1,2-Propylene Glycol Esters (Mixture of Regio- and Stereoisomers) | Ibuprofen Ethyl Ester | Ibuprofen Poly(oxy-1,2-ethanediyl) Ester | Ibuprofen 1,3-Butylene Glycol Esters (Mixture of Regio- and Stereoisomers) | Ibuprofen 2,3-Butylene Glycol Ester | Ibuprofen Impurity H | rac a-Hydroxy Ibuprofen | Ibuprofen Impurity G | Ibuprofen EP Impurity H | Ibuprofen impurity O | Ibuprofen EP Impurity F | Ibuprofen impurity at RRT 1.93 | Ibuprofen Sorbitol Ester (Mixture of Diastereomers) | Ibuprofen Ester Impurity |